Laquinimod
Laquinimod är en first-in-class immunmodulator med en ny verkningsmekanism i utveckling för behandling av svåra inflammatoriska ögonsjukdomar, såsom icke-infektiös uveit.
Detta är laquinimod
I experimentella modeller för autoimmuna/inflammatoriska sjukdomar har det visats att laquinimod aktiverar aryl hydrocarbon-receptorn (AhR) som finns i antigenpresenterande celler och är involverad i regleringen av dessa celler. Genom att påverka AhR omprogrammeras antigenpresenterande celler till att bli tolerogena, så i stället för att aktivera T-celler som orsakar inflammation aktiveras de regulatoriska T-cellerna med anti-inflammatoriska egenskaper, vilket i sin tur leder till att inflammationen dämpas.

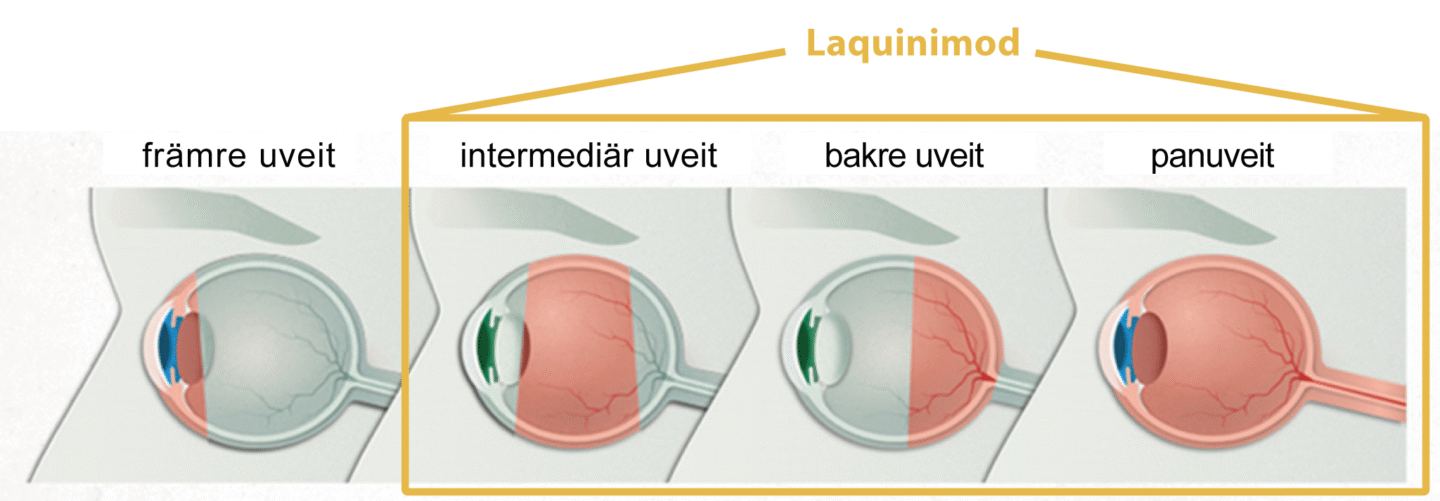
Icke-infektiös uveit
Icke-infektiös uveit är ett samlingsnamn för inflammationer i ögats druvhinna (uvea). Hit räknas iris, ciliarkropp och åderhinna. Uveit kan också leda till inflammationer i intilliggande vävnader, såsom näthinnan, den optiska nerven och glaskroppen, i frånvaro av en smittsam orsak. Uvea är avgörande för tillförsel av syre och näringsämnen till ögonvävnaden, och inflammation i uvea kan orsaka allvarliga vävnadsskador i ögat som kan leda till allmänna synproblem och risk för blindhet. Därutöver är vanliga symptom fläckar i synfältet, smärta i ögonen och röda ögon, ljuskänslighet, huvudvärk, små pupiller och förändrad färg på iris.
Om uveit inte behandlas kan det leda till allvarliga ögonproblem såsom blindhet, starr, glaukom, skador på den optiska nerven och näthinneavlossning. Icke-infektiös uveit uppstår ofta i samband med systemiska autoimmuna sjukdomar som sarkoidos, multipel skleros och Crohns sjukdom.
Uveit kan delas in i undertyper beroende på lokaliseringen av inflammationen. Intermediär, bakre och panuveit (icke-anteriör icke-infektiös uveit, NA-NIU) är de allvarligaste och mycket återkommande formerna som kan orsaka blindhet om de inte behandlas. Laquinimod utvecklas som ett nytt behandlingsalternativ för icke-infektiös uveit.
Marknad
Det finns begränsade behandlingsalternativ för patienter med icke-infektiös uveit. Den behandling som flertalet patienter genomgår är långtidsbehandling med höga doser kortikosteroider. Fortfarande uppnår cirka 40 procent av patienterna inte sjukdomskontroll, alternativt kan inte fortsätta med höga doser kortikosteroider på grund av biverkningar (Rosenbaum JT. Uveitis: treatment. In: Post TW, ed. UpToDate. Waltham (MA): UpToDate; 2021).
På senare tid har intraokulära injektioner med kortikosteroider introducerats med positiva effekter för vissa patienter och med begränsade systemiska kortikosteroid-relaterade biverkningar. Att injicera en depå med fördröjd frisättning av kortikosteroider i ögat förknippas med risker som grå starr och ökat intraokulärt tryck.
Den globala försäljningen av läkemedel för behandling av uveit uppgick 2023 till cirka 522 miljoner USD och försäljningen förväntas öka till cirka 1,5 miljarder USD år 2033 (Global Data Report March 2025, Uveitis – Opportunity Assessment and Forecast).
Det finns en betydande möjlighet för laquinimod inom segmentet icke‑infektiös, icke‑främre uveit, med cirka 550 000 behandlingsbara patienter och en marknadspotential på 1,5 miljarder USD.
Befintliga behandlingar
Standardbehandling idag för patienter med icke-infektiös uveit är höga doser med orala kortikosteroider eller injektioner av kortikosteroid i eller runt ögat. Immunsuppressiva medel, såsom metotrexat eller cyklosporin, används som kortikosteroid-sparande regim i andra behandlingslinjen, medan anti-TNF-antikroppar (Humira) används som andra eller tredje behandlingslinje.
Det finns ett stort medicinskt behov av nya effektiva och säkra terapier för icke-infektiös icke-anteriör uveit:
- cirka 35 procent av patienterna lider av allvarliga synproblem med risk för blindhet
- cirka 40 procent av patienterna svarar inte på behandling med kortikosteroider
- långtidsbehandling med höga doser kortikosteroider är förenat med allvarliga biverkningar
- för närvarande finns ingen topikal behandling tillgänglig
Det finns således ett behov av nya behandlingar med kompletterande effekt till kortikosteroider för att begränsa antalet patienter som inte svarar på behandlingar i första linjen. Dessutom finns det ett behov av säkrare terapier som kan reducera eller ersätta långtidsanvändning av kortikosteroider samt en behandling som kan administreras topikalt och nå ögats bakre delar för att minimera systemiska biverkningar och injektionsrelaterade risker.
Laquinimod för icke-infektiös uveit
Laquinimod kommer att utvecklas som en ny behandling för icke-infektiös uveit och har potential att användas i behandlingslinje 1 som ett tillägg till kortikosteroider men även i behandlingslinje 2 för patienter som inte svarat på behandling med kortikosteroider.
Klinisk utveckling
En innovativ ögondroppsformulering av laquinimod har utvecklats och ett prekliniskt säkerhets- och toxicitetsöverbryggande program för topikal behandling har slutförts. En klinisk fas I studie har genomförts för att dokumentera säkerhet och tolerabilitet av ögondroppsformuleringen i friska försökspersoner.
För att säkerställa att laquinimod når de bakre delarna i ögat inför vidare utveckling inom icke-anteriör uveit, har en klinisk biodistributionsstudie genomförts. Top-lineresultaten från LION-studien (Safety, Tolerability, and Distribution of Topical Laquinimod Eye Drops, an Innovative ImmunomodulatOr Targeting Aryl HydrocarboN Receptor) visar att dagliga doser av antingen 0,6, 1,2 mg och 1,8 mg resulterade i dosrelaterade intraokulära koncentrationer av laquinimod, som nådde en terapeutiskt relevant nivå i både glaskroppen och främre kammaren. Laquinimod administrerat som ögondroppar vid de valda dagliga dosnivåerna var säker och väl tolererad under den studerade administreringsperioden, och inga dosbegränsande toxiciteter rapporterades hos någon av deltagarna.
LION studien presenterades vid det största vetenskapliga mötet inom fältet för uveit och ögoninflammation, International Ocular Inflammation Society (IOIS) Congress, i Rio de Janeiro, Brasilien, den 26:e juni, 2025 och vid American Academy of Ophthalmology (AAO) årliga möte i oktober 2025.
Studien har genomförts i samarbete med forskare vid Byers Eye Institute, Stanford University (Palo Alto, Kalifornien, USA) med huvudprövare Quan Dong Nguyen, MD, MSc, FAAO, FARVO, FASRS, professor i oftalmologi, medicin och pediatrik, Stanford University School of Medicine.
Mer information om studierna finns i rutan nedan och på www.clinicaltrials.gov.
En klinisk fas II-studie med ögondroppsformulering av laquinimod i patienter med icke-infektiös uveit är planerad. Start av studien är villkorat ett partnersamarbete.
Tidigare klinisk erfarenhet med laquinimod
Under åren av sen klinisk produktutveckling genererades data avseende klinisk effekt och säkerhet för oral laquinimod i fler än 5 000 patienter, främst i multipel skleros (MS)-patienter, vilket motsvarar över 14 000 patient-år av exponering. Omfattande dataset inklusive regulatoriskt paket med preklinisk och klinisk säkerhet och CMC-dokumentation i full kommersiell skala har genererats.
